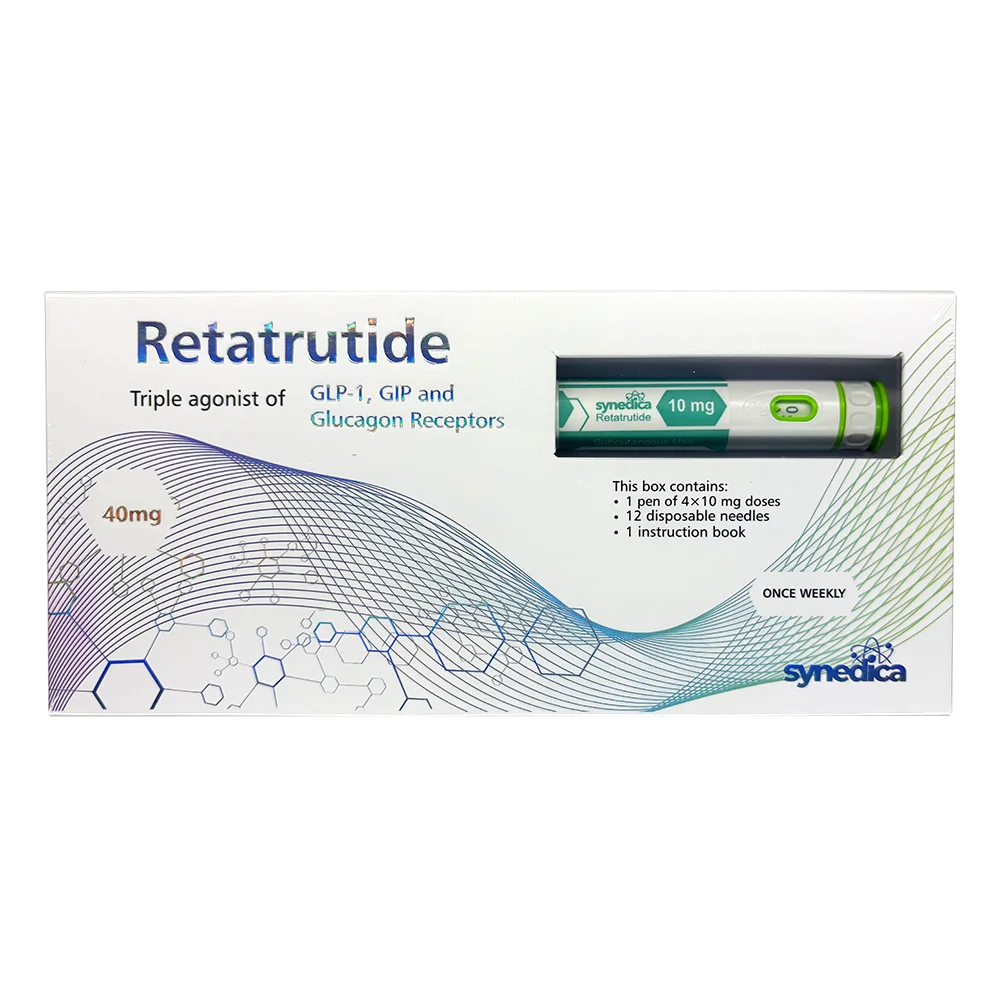
Synedica Retatrutide 40mg
£170.00
Synedica Retatrutide 40mg. A research-grade injectable kit called the Synedica Retatrutide 40mg Pen was created to investigate sophisticated weight-loss mechanisms via triple receptor agonism (GLP-1, GIP, and glucagon).
Synedica Retatrutide 40mg Pen | Synedica Retatrutide 40mg
Synedica Retatrutide 40mg Pen, Synedica Retatrutide 40mg. A research-grade injectable kit called the Synedica Retatrutide 40mg Pen was created to investigate sophisticated weight-loss mechanisms via triple receptor agonism (GLP-1, GIP, and glucagon). Although it is one of the most advanced instruments in metabolic research, it is not authorized for ingestion by humans or animals.
Synedica Retatrutide 40mg Pen Overview
- Producer: Synedica Laboratories
- Goal: Drug formulation, peptide stability, and metabolic science research and development (R&D).
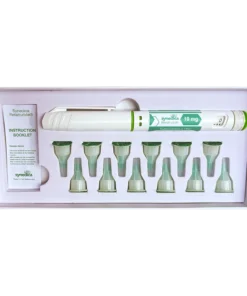
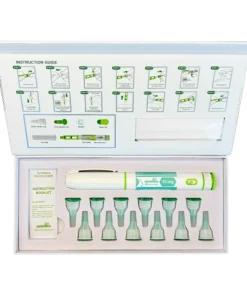
- Contents: One injectable pen with a 40 mg capacity, twelve disposable needles, and an instruction manual are included in each kit.
- Dosage Options: There are four complete doses of 10 mg each, and the pen offers variable dosing in increments of 2.5 mg, 5 mg, 7.5 mg, or 10 mg.
Synedica Retatrutide 40mg Pen | Mechanism of Action
Retatrutide is a synthetic peptide that controls metabolism and appetite by imitating natural hormones. It differs from earlier substances like tirzepatide or semaglutide due to its distinct triple receptor agonism:
- GLP-1 receptor activation: Promotes satiety and reduces caloric intake.
- GIP receptor activation: Enhances GLP-1 effects and may reduce gastrointestinal side effects.
- Glucagon receptor activation: Enhances fat breakdown, controls hepatic glucose production, and raises energy expenditure.
Applications of Research
The Synedica Retatrutide 40 mg Pen is not intended for clinical usage but rather for controlled laboratory research. Important uses consist of:
- Assessing peptide degradation under oxidative, enzymatic, and thermal stress is known as stability testing.
- Enhancing solubility, bioavailability, and structural integrity through formulation development.
- Testing interactions with inactive components is known as excipient compatibility.
- Protease Resistance: Evaluating resistance to enzymatic degradation.
- Encapsulation Studies: Controlling release with liposomes or microspheres.
- Administration Route Assessment: Investigating in vitro, dermal, and injectable delivery techniques.
Results of the Research
Retatrutide phase 2 trials have demonstrated impressive weight loss outcomes in controlled environments.
- 1 mg dose: typical weight loss of about 8.7%.
- 4 mg dose: decrease of about 17.1%.
- 8 mg dosage: approximately 22.8% decrease.
- 12 mg dose: ~24.2% reduction, with 26% of subjects losing at least 30% of their body weight—a level that was previously thought to be unachievable with pharmacological intervention alone.
Important Lessons
- A state-of-the-art research instrument for examining metabolic control and weight-loss pathways is the Synedica Retatrutide 40 mg Pen.
- Its triple receptor agonism is more effective than previous incretin-based drugs.
- Although it shows promise in research, it is not an approved therapeutic product and is still only used for research and development.
Be the first to review “Synedica Retatrutide 40mg” Cancel reply
Related products
Synedica Retatrutide
Synedica Retatrutide
Synedica Retatrutide
Synedica Retatrutide
Synedica Retatrutide
Synedica Retatrutide









Reviews
There are no reviews yet.